The vaccine was made too quickly, so I don’t trust it to be safe.
Researchers have been working on developing vaccines against coronaviruses long before COVID was upon us. During COVID vaccine development, phases overlapped to speed up the process, but all 3 phases were still completed.
How a New Vaccine is Developed, Approved and Manufactured
The Food and Drug Administration (FDA) sets rules for the three phases of clinical trials to ensure the safety of the volunteers. Researches test vaccines with adults first.
PHASE 1
20-100 Healthy Volunteers
- Is the vaccine safe?
- Does the vaccine seem to work?
- Are there any serious side effects?
- How is the size of the dose related to side effects?
PHASE 2
Several Hundred Volunteers
- What are the most common short-term side effects?
- How are the volunteers’ immune systems responding to the vaccine?
PHASE 3: Long-Term Discovery
Hundreds or Thousands of Volunteers
- How do people who get the vaccine and people who do not get the vaccine compare?
- Is the vaccine safe?
- Is the vaccine effective?
- What are the most common side effects?
Normally takes 1-4 years to discover any rarer or long-term side effects, usually done on 300-3,000 volunteers. The COVID vaccine studies used 30,000-40,000 volunteers to speed up the ability to detect rare side effects.
After Phase 3, the FDA grants licensure and the drug or vaccine goes to market. Sometimes, Phase 4 trials are conducted, which are long term studies lasting years to continue monitoring the safety and efficacy.
COVID vaccines were granted “Emergency Use Authorization” after being studied for 3 months in phase 3 and now have full FDA approval after being studied for over 1 year.
Pfizer has full FDA approval in adults as of 8/2021, Moderna applied for full FDA approval, but is still pending (application submitted Aug 2021). J&J is expected to apply for full FDA approval in late 2021.
The FDA will license the vaccine only if it’s safe, effective and the benefits outweigh the risks.
The Moderna Vaccine Trial Design
Began with 30,420 participants, stratified by health risk and age (<65 or ≥65 years old) and randomized 1:1, meaning half of the participants received a placebo and the other half received the vaccine. Throughout the entire trial, there is frequent adverse event and symptom monitoring. 1 month later, participants received the second dose of the vaccine or another placebo depending on what they previously received. Ongoing surveillance continues for up to two years from 14 days to two years past second dose, per protocol. Monitoring includes occurrence of COVID-19 with or without symptoms for both groups.
Does the Moderna vaccine work?
Number of COVID-19 Cases (n/N)
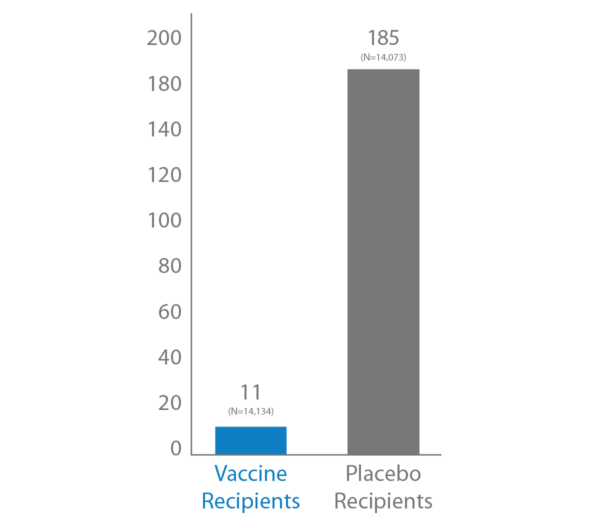
This chart shows the recorded number of trial participants who contracted COVID-19, with a median length of follow up being 9 weeks after the second dose.
Of the placebo recipients, 185 contracted the virus. Of the vaccinated recipients, only 11 contracted the virus.
Vaccine Efficacy† = 94.1% (95% CI, 89.3%, 96.8%) p<0.001‡
The Pfizer vaccine efficacy was very similar at 95%.
Cumulative COVID-19 Incidence
-
Placebo
-
mRNA-1273
This chart reports the cumulative COVID-19 incidence (CI) of vaccine and placebo recipients during the 120 days after their second dose. The incidence rate is measured per 1000 person-yr.
The placebo’s incidence rate was 79.7%, while the vaccinated population’s incidence rate was only 5.6%.
N Engl J Med 2021;384:403-16. DOI: 10.1056/NEJMoa2035389
Is the Moderna vaccine safe?
Most Common Adverse Effects (within 7 days of injection)
Adverse Events Reported Within 28 Days of Each Injection
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Unsolicited Adverse Event n (%)
Placebo Group
Vaccinated Group
Total
Deaths in Placebo Group:
Intraabdominal perforation
Cardiac arrest
SIRS from CLL
Deaths in Vaccine Group:
Cardiac arrest
Suicide
Aside from transient local and systemic side effects, no safety concerns were identified with COVID vaccine.
Don’t forget the research is still ongoing, plan is to monitor all trial volunteers for 2 years.
Based on what we know from our history of studying vaccines for over a century, most vaccine related adverse events occur within the first 6 weeks of administration. These initial trials looking at the first 1-2 months after vaccine administration are likely capturing most of the adverse events. However, we will continue to study them long term as we do with all vaccines.
N Engl J Med 2021;384:403-16. DOI: 10.1056/NEJMoa2035389
Ongoing Safety Monitoring for All COVID Vaccines
After a vaccine is approved and the public begins using it, several vaccine surveillance systems monitor for adverse events that may have not been detected during clinical trials: VAERS, VSD, CISA, CMS, BEST, V-SAFE. Learn more about CDC’s COVID vaccine monitoring programs.
The FDA and CDC closely monitory vaccine safety after the public begins using the vaccine.
The purpose of monitoring is to watch for adverse events (possible side effects). Monitoring a vaccine after it is licensed helps ensure that possible risks associated with the vaccine are identified.
Vaccine Adverse Event Reporting System (VAERS) | VAERS collects and analyzes reports of adverse events that happen after vaccination. Anyone can submit a report, including parents, patients and healthcare professionals.
Vaccine Safety Datalink (VSD) and Post-Licensure Rapid Immunization Safety Monitoring (PRISM) | Two networks of healthcare organizations across the U.S. VSD can analyze healthcare information from over 24 million people. PRISM can analyze healthcare information from over 190 million people. Scientists use these systems to actively monitory vaccine safety.
Clinical Immunization Safety Assessment Project (CISA) | CISA is a collaboration between CDC and 7 media research centers. Vaccine safety experts assist U.S. healthcare providers with complex vaccine safety questions about their patients. CISA conducts clinical research studies to better understand vaccine safety and identify prevention strategies for adverse events following immunization.
Vaccine recommendations may change if safety monitoring reveals new information on vaccine risk, like if scientists detect a new serious side effect.
For more information, read about Vaccine Safety.
As of November 8, 2021, over 432 million doses of COVID-19 vaccine have been given in the U.S. Below are examples of other vaccine’s symptoms for reference:
Anaphylaxis: Applies to any COVID-19 vaccine. Chances are 2.5-11.1 in 1 million
Thrombosis with Thrombocytopenia (TTS): J&J only. Chances are 7 in 1 million women aged 1-49
Myocarditis/Pericarditis: Moderna/Pfizer. Chances are 40.6 in 1 million males aged 12-29
https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/safety-of-vaccines.html
COVID-19 isn’t that serious. I’m young and healthy, so I’ll take my chances
COVID-19 was the number 1 leading cause of death for people ages 35-54 years in September 2021. It felt to rank #7 among leading causes of death in July, but is back up to #2 as of September, 2021. Source
Based on CDC reports, an estimate of 98.6% of people hospitalized with COVID-19 between June and August 2021 were unvaccinated. Over 280,000 COVID-19 hospitalizations could have been prevented by vaccination between June to August 2021. Conservative estimates indicate an average COVID-19 hospitalization costs about $20,000. This means preventable COVID-19 hospitalizations cost $5.7 billion from June to August in 2021. Source
Well, I already had COVID, so I don’t need the vaccine.
Evidence is emerging that people get better protection by being fully vaccinated compared with having had COVID-19.
Cavanaugh et al. (MMWR Aug 2021) showed that unvaccinated people who already had COVID-19 are more than 2 times as likely than fully vaccinated people to get COVID-19 again.
I’m pregnant, so I can’t get the vaccine. Afterwards, I’m breastfeeding, so I still can’t get the vaccine.
ACOG recommends all eligible persons >12 years, including pregnant and lactating individuals, receive a COVID-19 vaccine.
No evidence of any adverse maternal or fetal effects from COVID-19 vaccination in pregnant or lactating individuals. (Ciapponi 2021, Wainstock 2021, Kachikis 2021, Magnus 2021, Bertrand 2021) Pregnant and recently pregnant patients with COVID-19 are at increased risk of more severe illness. (Ellington MMWR 2020, Collin 2020, Delahoy MMWR 2020, Khan 2021)
ACOG recommends pregnant and recently pregnant women (up to 6 wk postpartum) receive a booster dose of COVID-19.
If you had J&J vaccine, get booster at least 2 months after initial vaccine. If you had mRNA vaccine, get booster at least 6 months after initial vaccine.
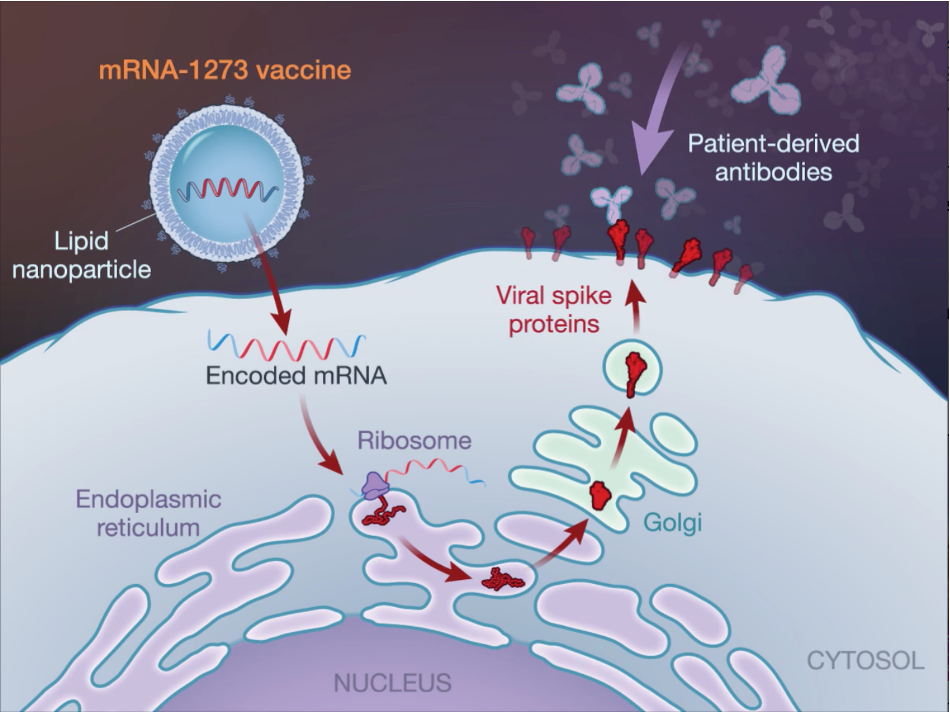
COVID-19 mRNA vaccines might alter my DNA, I’m not putting that in my body.
COVID-19 mRNA vaccines cannot give someone the virus
They do not affect or interact with our DNA in any way
The mRNA and the spike protein don’t last long in the body
mRNA vaccines are newly available to the public but have been studied for decades
COVID vaccines are much safer than a COVID infection
COVID vaccines are safe. Aside from transient side effects immediately after injection, there is very low risk of any serious adverse events.
COVID vaccines have been thoroughly tested and have passed all the FDA’s rigorous safety requirements
COVID-19 virus poses a real threat causing hundreds of thousands of deaths and hospitalizations, the majority of which could be prevented by vaccination
COVID vaccination provides better protection than simply having had COVID-19 infection
There is no evidence that COVID vaccines cause issues with pregnancy, lactation, or infertility
mRNA vaccine technology has been around for decades and is safe
Helpful Resources
https://www.cdc.gov/coronavirus/2019-ncov/vaccines/index.html
https://www.healthsystemtracker.org/archive/?_sft_post_tag=covid-19

